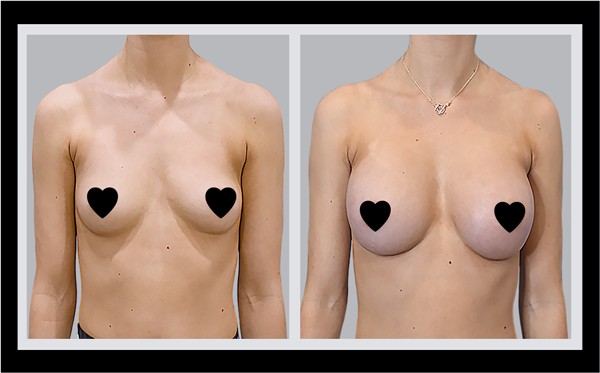
LIVE LARGE
BREAST AUGMENTATION
Breast Augmentation
Women come to Buckhead Plastic Surgery in Atlanta with diverse reasons for wanting breast augmentation. Some have wanted larger, shapelier breasts for many years and are realizing their dreams of feeling more confident about their bodies. Other patients have seen their breasts change as they got a little older, or had children, and want to regain the look they had when they were younger.
The reasons for breast augmentation are as unique as each patient, and double board-certified plastic surgeon Dr. Alan Larsen knows the decision is deeply personal. Due to his years of experience and widespread reputation for excellent breast augmentation results, Dr. Larsen routinely treats women from Decatur, Marietta, and throughout Georgia.
Request a consultation with Dr. Larsen to discuss your breast augmentation options using the online form, or call our office at (404) 367-9005 and a member of our staff will help you schedule an appointment.
![]()
“You get what you pay for and I definitely got what I paid for and more!
The staff was amazing. Dr. Larsen was very knowledgeable and I was comfortable with him performing the surgery since his reviews were the best and he is a top doctor. Can’t get any better than that!”*
*Your results may vary.
Read more reviews from some of Dr. Larsen’s actual patients.
![]()
Your Breast Augmentation Options
Choosing the size and shape of your breast implants can be an exciting, if sometimes overwhelming, part of your breast augmentation journey. But those are only some of the breast enhancement decisions you’ll make with the help of Dr. Larsen’s expertise. Choices involving the material filling the implants, the placement of incisions, and where Dr. Larsen will insert the implants are integral to getting the customized results you want.
You can see a 3D preview of the possible results using the Sculpt My Dream technology at our practice.
Choosing Breast Implants

By offering a range of breast implant choices, Dr. Larsen helps personalize your breast augmentation procedure. Silicone gel implants are the most popular choice among patients at Buckhead Plastic Surgery and throughout the world. Most of our patients believe that the latest generation of silicone gel implants look and feel more like natural breast tissue than saline implants.

LEARN MORE ABOUT
BREAST SURGERY
How Much is a Breast Augmentation in Atlanta?
Understanding Your Breast Implant Options
20 Questions to Ask Your Breast Augmentation Surgeon
What to Expect During Breast Augmentation Recovery
What You Should Know About Breast Reconstruction
The Best Breast Augmentation Plastic Surgeon in Atlanta
The Essential Guide to Breast Implants
What to Know About Exercise After Breast Augmentation
The Myths About Silicone Breast Implants
The Pros and Cons of Fat Transfer Breast Augmentation
Personality Factors Affect Quality of Life After Breast Reconstruction
At Buckhead Plastic Surgery we are proud to offer Natrelle Implants
- Natrelle Silicone Implant (often called “gummy bear” implants) are only available for purchase by double board-certified plastic surgeons.
- Natrelle offers 5 different profiles in 3 different gummy gels with 300 options
The cohesive silicone gel, used to fill breast implants isn’t like a liquid at all. It maintains its shape, but is remarkably pliable and closely resembles the feel of natural breast tissue. Cohesive silicone gel breast implants maintain their shape – even when they’re cut in half.
We also offer saline breast implants for patients who do not want silicone implants or for young women under the age of twenty-two, as regulated by the FDA. One reason a patient may choose saline implants is because, unlike silicone implants which arrive pre-filled, Dr. Larsen fills saline implants after inserting them. That means the incisions are slightly smaller than those for silicone implants. They are also less expensive than their silicone counterparts.
Your Procedure
Dr. Larsen creates a customized surgical plan after discussing your cosmetic goals and conducting a physical examination during your consultation. That plan involves the incision location and where Dr. Larsen creates the implant pocket. Saline and silicone implants can be placed through incisions in the crease below the breast, around the areola, or in the armpit. The pocket can be located either behind the breast tissue (subglandular), or beneath the pectoral muscle (submuscular).
You may also benefit from a Strattice™ internal bra, which is an innovative device Dr. Larsen carefully places within the implant pocket that can help prevent visible rippling of your breast implants. This internal bra procedure minimizes any skin stretching associated with enhancing the size of your breasts and supports the implant to promote longer-lasting breast augmentation results.
Your Breast Augmentation Options
Choosing the size and shape of your breast implants can be an exciting, if sometimes overwhelming, part of your breast augmentation journey. But those are only some of the breast enhancement decisions you’ll make with the help of Dr. Larsen’s expertise. Choices involving the material filling the implants, the placement of incisions, and where Dr. Larsen will insert the implants are integral to getting the customized results you want.
You can see a 3D preview of the possible results using the Sculpt My Dream technology at our practice.
Choosing Breast Implants

By offering a range of breast implant choices, Dr. Larsen helps personalize your breast augmentation procedure. Silicone gel implants are the most popular choice among patients at Buckhead Plastic Surgery and throughout the world. Most of our patients believe that the latest generation of silicone gel implants look and feel more like natural breast tissue than saline implants.
At Buckhead Plastic Surgery we are proud to offer the Natrelle Implants
- Natrelle Silicone Implant (often called “gummy bear” implants) are only available for purchase by double board-certified plastic surgeons.
- Natrelle offers 5 different profiles in 3 different gummy gels with 300 options

The cohesive silicone gel, used to fill breast implants isn’t like a liquid at all. It maintains its shape, but is remarkably pliable and closely resembles the feel of natural breast tissue. Cohesive silicone gel breast implants maintain their shape – even when they’re cut in half.
We also offer saline breast implants for patients who do not want silicone implants or for young women under the age of twenty-two, as regulated by the FDA. One reason a patient may choose saline implants is because, unlike silicone implants which arrive pre-filled, Dr. Larsen fills saline implants after inserting them. That means the incisions are slightly smaller than those for silicone implants. They are also less expensive than their silicone counterparts.
Your Procedure
Dr. Larsen creates a customized surgical plan after discussing your cosmetic goals and conducting a physical examination during your consultation. That plan involves the incision location and where Dr. Larsen creates the implant pocket. Saline and silicone implants can be placed through incisions in the crease below the breast, around the areola, or in the armpit. The pocket can be located either behind the breast tissue (subglandular), or beneath the pectoral muscle (submuscular).
You may also benefit from a Strattice™ internal bra, which is an innovative device Dr. Larsen carefully places within the implant pocket that can help prevent visible rippling of your breast implants. This internal bra procedure minimizes any skin stretching associated with enhancing the size of your breasts and supports the implant to promote longer-lasting breast augmentation results.
Recovery
Breast augmentation is an outpatient procedure, meaning you return home the same day as the surgery is performed. You’ll need to arrange for someone to drive you home and care for you for the first few hours after your surgery. It typically takes about 3 to 5 days before you can return to work, depending on the physical nature of your job. Many women schedule breast augmentation on a Thursday and are able to go back to work the following Monday.
It will take at least 2 weeks before resuming certain physical activities, and patients who have implants placed beneath the chest muscles require a longer recovery. Dr. Larsen provides all patients with detailed postsurgical instructions that, when followed, help the healing process go more smoothly.
Breast Implant Cost:
Starting at $8,000.00
Recovery
Breast augmentation is an outpatient procedure, meaning you return home the same day as the surgery is performed. You’ll need to arrange for someone to drive you home and care for you for the first few hours after your surgery. It typically takes about 3 to 5 days before you can return to work, depending on the physical nature of your job. Many women schedule breast augmentation on a Thursday and are able to go back to work the following Monday.
It will take at least 2 weeks before resuming certain physical activities, and patients who have implants placed beneath the chest muscles require a longer recovery. Dr. Larsen provides all patients with detailed postsurgical instructions that, when followed, help the healing process go more smoothly.
Breast Implant Cost:
Starting at $8,000.00
Common Questions
How much does breast augmentation cost?
At Buckhead Plastic Surgery, the cost of breast augmentation starts at $8,000.00. The final price depends on several factors, including the type of breast implants you choose and the extent of the procedure. As noted above, cohesive silicone gel implants are a bit more expensive than saline implants.
We believe in helping qualified patients achieve their cosmetic goals. That’s why we offer help with financing for breast augmentation and other aesthetic procedures. We work with several financing companies who specialize in providing loans for healthcare services.
Is it best to have implants placed under or over the muscle?
This decision is best left until after you have discussed the benefits of both choices with Dr. Larsen during the consultation. A woman with little existing breast tissue usually gets better results by placing implants under the muscle, which helps create a more natural look. Patients with more significant breast tissue may opt for having implants placed above the muscle. There are other factors to consider, including your fitness habits, for example, or whether you want more fullness in the upper portion of the breasts.
I’ve heard of “natural” breast augmentation. What is that?
The fat transfer technique can add volume to the breasts without implants. Fat transfer, sometimes also called fat grafting, uses fat from other areas of your body to add volume to the breasts. The fatty tissue is extracted using liposuction, processed to remove impurities, and then carefully re-injected into the breasts. The technique is best for patients who want only a modest increase in the size of their breasts.
Should I choose round or shaped implants?
As with other choices involved with breast augmentation, this one depends primarily on your cosmetic goals. The anatomically shaped silicone gel implants mimic the breasts’ natural slope, with more fullness in the lower two-thirds. Some patients favor that look, while those who choose round implants generally prefer to increase the fullness in the upper portion of their breasts.
Common Questions
How much does breast augmentation cost?
At Buckhead Plastic Surgery, the cost of breast augmentation starts at $8,000.00. The final price depends on several factors, including the type of breast implants you choose and the extent of the procedure. As noted above, cohesive silicone gel implants are a bit more expensive than saline implants.
We believe in helping qualified patients achieve their cosmetic goals. That’s why we offer help with financing for breast augmentation and other aesthetic procedures. We work with several financing companies who specialize in providing loans for healthcare services.
Is it best to have implants placed under or over the muscle?
This decision is best left until after you have discussed the benefits of both choices with Dr. Larsen during the consultation. A woman with little existing breast tissue usually gets better results by placing implants under the muscle, which helps create a more natural look. Patients with more significant breast tissue may opt for having implants placed above the muscle. There are other factors to consider, including your fitness habits, for example, or whether you want more fullness in the upper portion of the breasts.
I’ve heard of “natural” breast augmentation. What is that?
The fat transfer technique can add volume to the breasts without implants. Fat transfer, sometimes also called fat grafting, uses fat from other areas of your body to add volume to the breasts. The fatty tissue is extracted using liposuction, processed to remove impurities, and then carefully re-injected into the breasts. The technique is best for patients who want only a modest increase in the size of their breasts.
Should I choose round or shaped implants?
As with other choices involved with breast augmentation, this one depends primarily on your cosmetic goals. The anatomically shaped silicone gel implants mimic the breasts’ natural slope, with more fullness in the lower two-thirds. Some patients favor that look, while those who choose round implants generally prefer to increase the fullness in the upper portion of their breasts.
Breast Lift vs Breast Augmentation – Find What’s Right For You
Breast augmentations and breast lifts are two popular breast surgeries that can help you boost your confidence. Whether you’re experiencing drooping breasts from [...]
Can I Receive a Breast Lift Without Implants?
For many women, a breast lift can do wonders to reclaim self-confidence. There’s just something delightful about high, round, perky breasts. This can [...]
Total Mom Makeover
Pregnancy and giving birth are one of the most miraculous transformations in nature. However, the process can take quite a toll on a [...]
How long do breast implants last?
Many women consider breast implants for various reasons, but did you know that breast implants don’t last forever? Although they do last a [...]
Patient Photo Gallery
The before-and-after photos of Dr. Larsen’s actual patients showcase the kind of results he consistently achieves.
Patient Video Gallery
Click here to watch our patient’s inspiring stories, testimonials, procedures and treatments from Buckhead Plastic Surgery & LUX Med Spa.
Alan N. Larsen, MD.
Double Board-Certified Atlanta Plastic Surgeon
Dr. Larsen established Buckhead Plastic Surgery as a boutique practice to offer world-class, personalized service to women and men throughout the Southeast. Dr. Larsen’s compassion and talent are complemented by his elite training with some of the nation’s finest plastic surgeons.
Alan N. Larsen, MD.Double Board-Certified Atlanta Plastic SurgeonDr. Larsen established Buckhead Plastic Surgery as a boutique practice to offer world-class, personalized service to women and men throughout the Southeast. Dr. Larsen’s compassion and talent are complemented by his elite training with some of the nation’s finest plastic surgeons.